Group members
Group members
Thesis defended
Mateusz Trochowski has got his PhD degree. Congratulations!
Diversity of the photosensitization mechanisms of surface-functionalized oxide semiconductors.
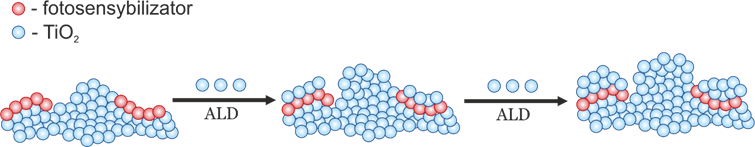
The doctoral thesis is focused on verification of four hypotheses related to the photosensitization mechanisms of titanium(IV) oxide and zinc oxide. The first hypothesis concerned the possibility of protection of photosensitizers bound to the TiO2 surface by covering them with a thin layer of titanium(IV) oxide to improve the photostability. The strategy worked only for a minority of the tested samples. The second hypothesis was the higher stability of dihydroxy derivatives of anthraquinone comparised to catechol adsorbed at TiO2. Long-term photocatalytic tests have shown that all obtained materials are more photostable than the reference catechol/TiO2 system and some of them are also more active. The third problem concerned investigation how insignificant modifications of photocatalysts can significantly change their photocatalytic activity. A significant effect on photocatalytic activity, of the studied materials, in particular upon visible light irradiation, was observed, although changes in the phase composition, spectroscopic properties and morphology of the samples tested were negligible. The fourth hypothesis concerned determining of the mechanisms of localized surface plasmon resonance interaction with ZnO modified with Au nanoparticles. The analysis of emission spectra has shown that photosensitization of ZnO occurs most probably via the transfer of “hot” electrons.